 There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. The grams to grams-moles (g mol) calculator is a valuable tool for anyone working with chemical calculations.  
Increases in temperature tend to decrease density since the volume will generally increase. To convert from moles to mass, we need the molar mass of bilirubin, which we can determine from its chemical formula: 33 C molar mass: 33 × 12.01 g. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. 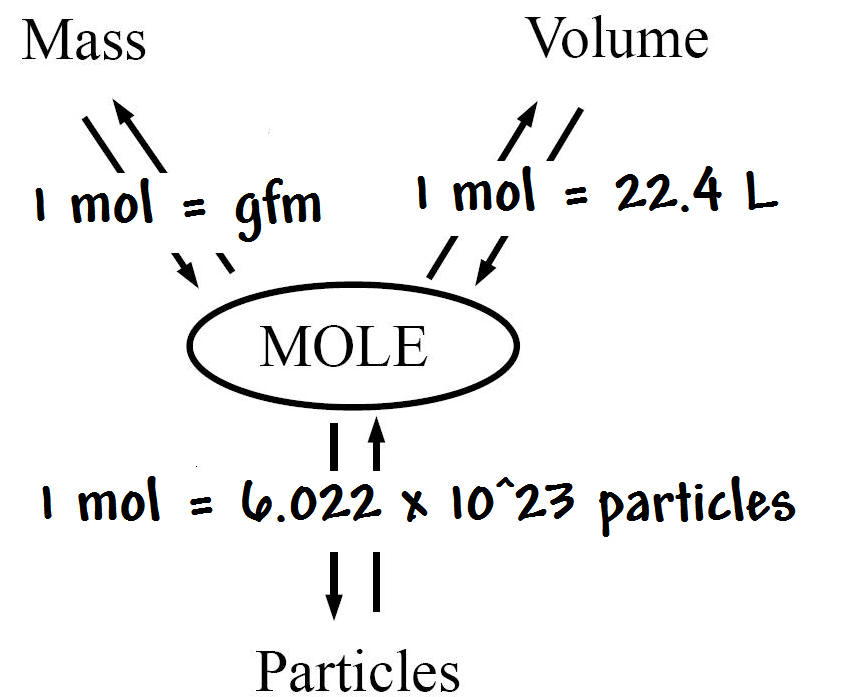
In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. The calculator will instantly calculate the results using the chemical equation for moles. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass Mole Calculator Select the chemical parameter (mass, moles, molecular weight) from the list and provide it in required ones. a pollution of 1 gram of benzene in a certain amount of water converts to N A /78.11 7. How much is 1 mole to grams The mass of one mole of a substance is equal to its molar mass in grams. Using the above calculator you could find that e.g. To calculate the number of moles of an element use the formula: n m/M, where m is the Mass of the element, and M is the Molecular weight. This calculator simplifies the process of determining the actual weight or quantity of. There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. Or 1 mole of a substance will contain Avogadros number of that substance. A Percent to Grams Calculator is a handy tool for converting percentages into gram values, especially when dealing with solutions, mixtures, or substances where the quantity of a particular component is expressed as a percentage of the total weight or volume. However, it is important to pay special attention to the units used for density calculations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
